- Blog
- Loading protein ladder for western blot transfer
- Pokemon super mystery dungeon decrypted rom download
- Linkedin logo hd png
- Online kink checklist
- English script font free
- Turn lightroom presets into capture one styles
- Mixed in key studio edition torrent
- Calorie tracker template
- Passport appointment usps scheduler
- Minecraft bedrock invincibility hack
- Windows downloading all icloud photo not originals
- Free widescreen powerpoint templates infographic
- Sample response to bill of particulars
- Powerpoint design ideas animation
- Free spider solitaire offline game
- Pokemon super mystery dungeon decrypted rom english
- Christiana Barkley charles barkley daughter instagram
- Google webstore chrome remote
- Ancient curse glyphs
- Btd5 serial key free
- X ray of dogs cervical spine
- Checksoft free download mac
- Facebook data extractor software crack india
- Movie productions companies in pittsburgh pa
- Arturia analog lab 4 system requirements
- Ghost of tsushima map of izuhara
- Free blank wheel of life template pdf
- Fortnite save thecworld redeem code xbox one
- Weight loss tracker 2019 template
- Deadbolt lock with key
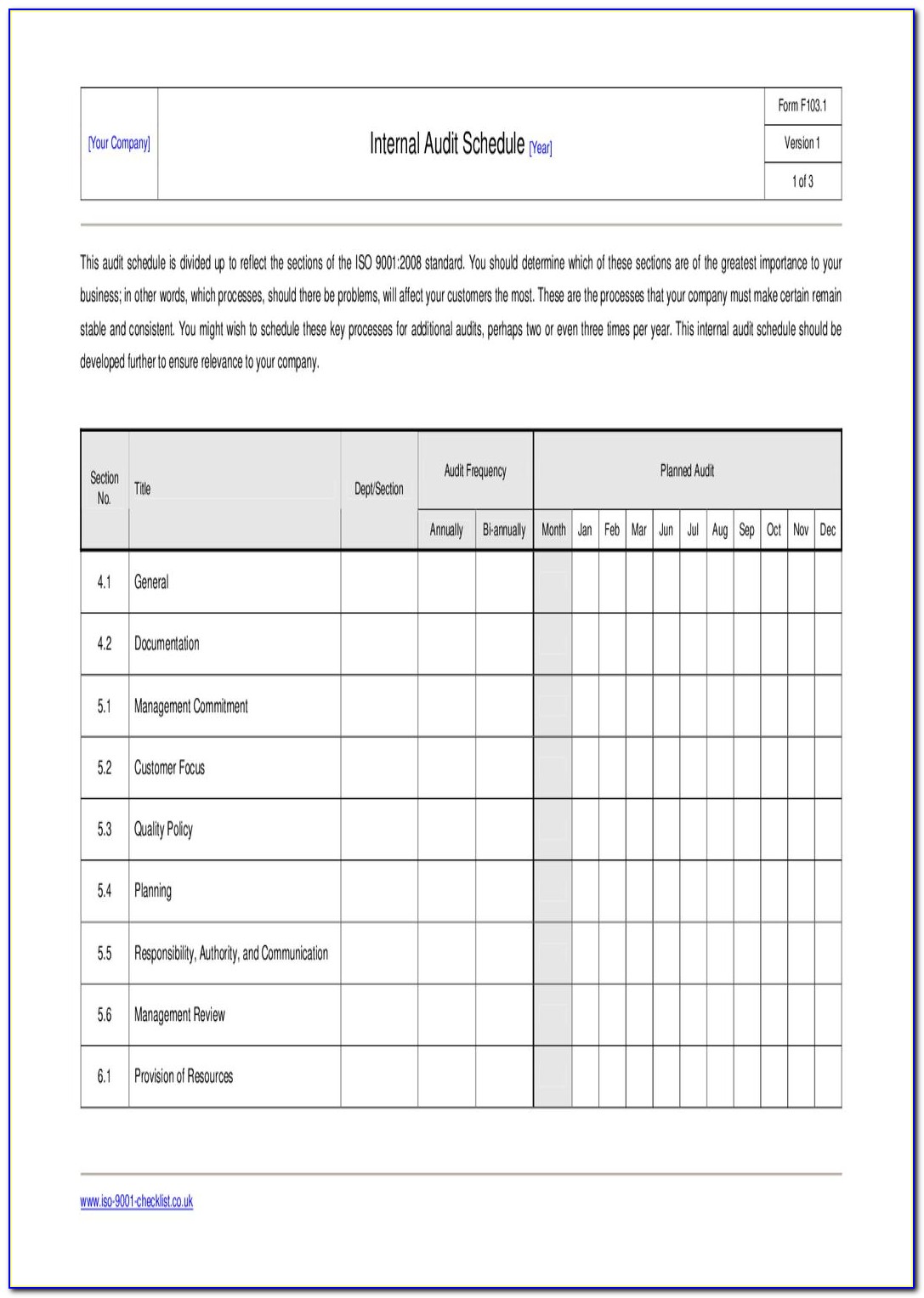
- Iso 13485 internal audit checklist
- Schedule passport at usps
- Download driver dvd windows media player
- Ultimate epic battle simulator full free download
- Free pogo spades online games
- Among us imposter logo
- Cursive font tattoo designs
- Gtr evolution download
- Car park maintenance checklist
- Free propresenter worship background
- Gta 5 apk download gratis pc
- Temple run oz game download apk
- Watch tv live internet spectrum
- Harvard resume template
- Bookmark folder icon color microsoft edge
- Joe biden first wife picture
- Maple tree types by leaf
- Shortcut for bullet point powerpoint
- Minecraft earth map download bedrock

Many companies seek the certification because of the financial benefits to their business. The Benefits of ISO 13485:2016īusinesses that have implemented ISO 13485 cite numerous benefits. For those seeking their first ISO 13485 certification, the following information can serve as a starting place for implementation. The auditing process can appear overwhelming, but it does not have to be. It is important for these organizations to understand the advantages of becoming ISO certified, identify the key differences between the 2003 publication and the 2016 publication and begin the work for first-time certification. If your company was once certified to ISO 13485:2003, and you want to reenter markets that require certification to ISO 13485, you may seek a first-time certification to ISO 13485:2016. Those that haven't have had to pull out of European Union (EU) and Canadian markets. Many organizations that needed to keep an active ISO 13485 certification have already adopted the 2016 standards. The three-year transition period from ISO 13485:2003 to ISO 13485:2016 ended in March 2019. Becoming ISO 13485 certified can increase your client base and reduce barriers to entry of foreign markets, product liabilities and production down-time. It assures a commitment to quality and increases efficiencies within the organization. ISO 13485 provides a great advantage for organizations producing medical devices and related services. ISO 13485 was developed to address these needs.
However, medical device and pharmaceutical companies have specialized requirements that made some of the requirements of ISO 9001 difficult to apply. ISO 13485 derived from ISO 9001, a quality management system certification that's available to businesses in a wide variety of industries. The most recent update to the standard was published in February of 2016, overriding previously published versions from 20. Ability to trace and recall products and devices.It's published by the International Organization for Standardization. ISO 13485 addresses: The ISO 13485 standard governs quality management systems for medical devices and related services. Clause 8: Measurement, Analysis & Improvement.Key Differences between ISO 13485 and ISO 9001.

At NQA, we have extensive experience with ISO 13485 and other standards relevant to the medical device manufacturing sector. One of these standards is ISO 13485, a quality management system (QMS) standard designed specifically for medical device manufacturers. To ensure the appropriate level of quality control, it's essential to comply with the relevant standards. Consistent product quality is an issue of patient safety, regulatory compliance and a company's ability to succeed in the industry. Product conformity is important across all industries, but it's especially crucial when it comes to the design and manufacturing of medical devices. We are always looking for talented people to join our team and work with a variety of interesting clients worldwide. Choose a certification body that has your best interests in mind. The NQA leadership team is made up of experts across a host of standards and industries. Take a look at some of the many industry associates and regulators we work alongside behind the scenes. NQA is involved in a number of technical committees. Improve your organisation and meet the requirements of your chosen standard with NQA.
We believe that clients deserve value for money and great service. That’s why our policy is to achieve accreditation for services wherever possible. NQA provides certification, training and support services to help improve processes, performance and products throughout your organisation.Įnsuring the integrity of standards and rigour of the certification process is key. Our services will help you improve your green league ranking, estate management and cost efficiency. See why certification to ISO standards is one of the best investments a contractor can make. The global construction industry is lucrative and competitive. Implement best practices across the food and beverage industry with certification in food safety, health, environmental and quality management standards. We are one of the world's leading certification bodies for the aviation and aerospace industry, serving Lockheed, Boeing, Raytheon, NASA and many more.Ĭyber security is essential for every business, which is why we work with both start-ups and conglomerates on tailored risk-based management systems. Learn how we're supporting the global industry across the automotive supply chain. Economic and environmental factors have changed the way vehicles must perform.
- Blog
- Loading protein ladder for western blot transfer
- Pokemon super mystery dungeon decrypted rom download
- Linkedin logo hd png
- Online kink checklist
- English script font free
- Turn lightroom presets into capture one styles
- Mixed in key studio edition torrent
- Calorie tracker template
- Passport appointment usps scheduler
- Minecraft bedrock invincibility hack
- Windows downloading all icloud photo not originals
- Free widescreen powerpoint templates infographic
- Sample response to bill of particulars
- Powerpoint design ideas animation
- Free spider solitaire offline game
- Pokemon super mystery dungeon decrypted rom english
- Christiana Barkley charles barkley daughter instagram
- Google webstore chrome remote
- Ancient curse glyphs
- Btd5 serial key free
- X ray of dogs cervical spine
- Checksoft free download mac
- Facebook data extractor software crack india
- Movie productions companies in pittsburgh pa
- Arturia analog lab 4 system requirements
- Ghost of tsushima map of izuhara
- Free blank wheel of life template pdf
- Fortnite save thecworld redeem code xbox one
- Weight loss tracker 2019 template
- Deadbolt lock with key
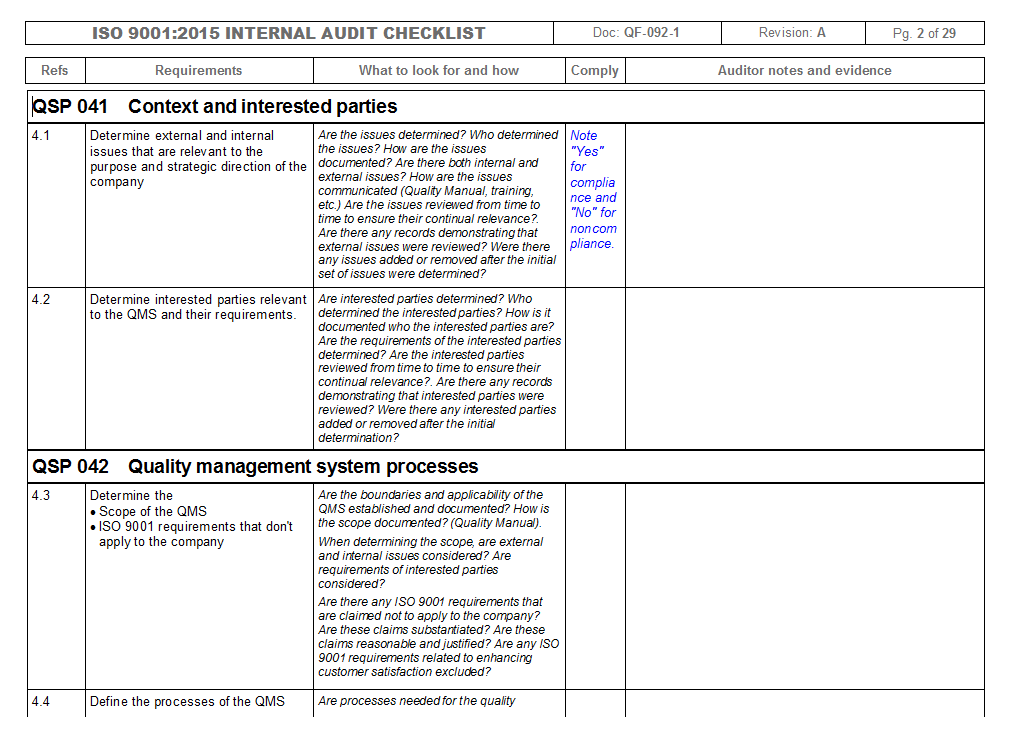
- Iso 13485 internal audit checklist
- Schedule passport at usps
- Download driver dvd windows media player
- Ultimate epic battle simulator full free download
- Free pogo spades online games
- Among us imposter logo
- Cursive font tattoo designs
- Gtr evolution download
- Car park maintenance checklist
- Free propresenter worship background
- Gta 5 apk download gratis pc
- Temple run oz game download apk
- Watch tv live internet spectrum
- Harvard resume template
- Bookmark folder icon color microsoft edge
- Joe biden first wife picture
- Maple tree types by leaf
- Shortcut for bullet point powerpoint
- Minecraft earth map download bedrock
